Investigating the molecular mechanism of HIF in hypoxic stress
The top three causes of death in Japan are malignant tumor, ischemic heart disease, and cerebral vascular disease. A common cause of these diseases is low oxygen tension in our body, known as hypoxic stress. The key transcription factors that regulate response to hypoxic stress are the Hypoxia Inducible Factors (HIFs). There are 3 types of HIFs: HIF-1α are expressed ubiquitously, HIF-2α are mainly expressed in endothelial cells, and HIF-3α works as an inhibitor against HIF-1α and HIF-2α.
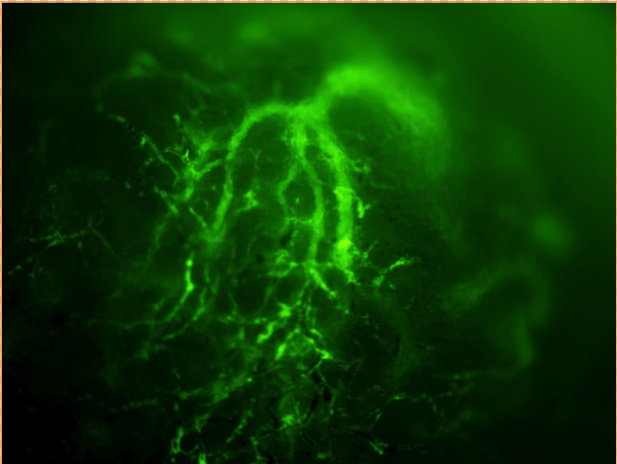
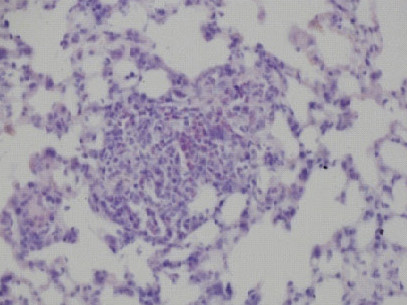
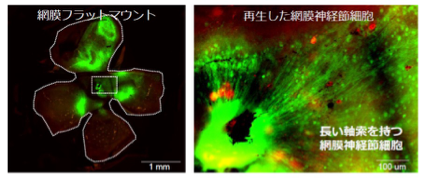
In our laboratory, we investigate the molecular mechanisms of HIFs for hypoxic responses in vivo. We investigate the roles of HIF-2α for tumor vascularization, erythropoiesis, retinopathy of prematurity using HIF-2α knockdown mice. We also analyze the roles of HIF-3α for pulmonary hypertension and lung development using HIF-3α knockout mice. We try to clarify the diversity of HIF functions in response to various hypoxic stimuli.