Intergrated BioMedical Science
Cell Dynamics
The Cell Dynamics division studies the roles of transforming growth factor-β-related molecules (TMEPAI, MAFK/GPNMB, THG-1) in tissue maintenance and cancer formation, especially in cancer stem cell biology.
Based on our novel findings how these molecules work to increase the number of cancer stem cells, we are aiming to establish novel cancer therapy and prevention methods using macrocyclic peptides targeting dormant cancer stem cells.
To achieve our research goal, we conduct multidisciplinary studies including molecular cell biology, pathological analysis of genetically modified mice, macrocyclic peptide screen-ing, three-dimensional quantitative tissue analysis, mathematical modeling, and protein structure analysis.
Related Research Papers
Research papers about GPNMB
Okita Y, Kimura M, Xie R, Chen C, Shen LTW, Kojima Y, Suzuki H, Muratani M, Saitoh M, Semba K, Heldin C-H, Kato M. The transcription factor MAFK induces EMT and malignant progression of triple-negative breast cancer cells through its target GPNMB. Science Signal. 10 (474), pii: eaak9397, 2017. doi: 10.1126/scisignal.aak9397. PMID: 28400538
Chen C, Okita Y, Watanabe Y, Abe F, Fikry MA, Ichikawa Y, Suzuki H, Shibuya A, Kato M. Glycoprotein nmb is exposed on the surface of dormant breast cancer cells and induces stem cell-like properties. Cancer Res. 78(22): 6424-6435, 2018. doi: 10.1158⁄0008-5472.CAN-18-0599.
Okita Y, Chen C, Kato M. Cell-surface GPNMB and induction of stemness. Oncotarget. 9(99):37289-37290, 2018. doi: 10.18632/oncotarget.26472. (Editorial)
Xie R, Okita Y, Ichikawa Y,Mohammed FA, Huynh Dam KT, Tran SPT, Kato M. Role of the kringle-like domain in Glycoprotein NMB for its tumorigenic potential. Cancer Sci. 110(7): 2237-2246, 2019. doi: 10.1111/cas.14076. PMID: 31127873
Research papers about PMEPA1/TMEPAI
Watanabe Y, Itoh S, Goto T, Ohnishi E, Inamitsu M, Itoh F, Satoh K, Wiercinska E, Yang W, Shi L, Tanaka A, Nakano N, Mommaas AM, Shibuya H, ten Dijke P and Kato M. TMEPAI, a transmembrane TGF-ß -inducible protein, sequesters Smad proteins from active participation in TGF-ß signaling. Mol Cell. 37: 123-134, 2010.
Nakano N, Itoh S, Watanabe Y, Maeyama K, Itoh F, and Kato M. Requirement of TCF7L2 for TGF-ß -dependent transcriptional activation of the TMEPAI gene. J Biol Chem. 285: 38023-38033, 2010.
Vo Nguyen TT, Watanabe Y, Shiba A, Noguchi M, Itoh S and Kato M. TMEPAI/PMEPA1 enhances tumorigenic activities in lung cancer cells. Cancer Sci. 105: 334-341, 2014. doi: 10.1111/cas.12355. Epub 2014 Feb 18. PMID:24438557.
Azami S, Vo Nguyen TT, Watanabe Y and Kato M. Cooperative induction of transmembrane prostate androgen induced protein TMEPAI/PMEPA1 by transforming growth factor-ß and epidermal growth factor signaling. (*equal contribution) Biochem Biophys Res Commun. 456: 580-585, 2015. doi: 10.1016/j.bbrc.2014.11.107. PMID: 25482449
Nakano N, Kato M, Itoh S. Regulation of the TMEPAI promoter by TCF7L2: the C-terminal tail of TCF7L2 is essential to activate the TMEPAI gene. J Biochem. 159: 27-30, 2016. doi:10.1093/jb/mvv117. PMID: 26590303.
Wardhani BWK, Puteri MU, Watanabe Y, Louisa M, Setiabudy R, Kato M. Knock-out transmembrane prostate androgen-induced protein gene suppressed triple-negative breast cancer cell proliferation. Medical J. Indonesia 26(3): 178-182, 2017.
Amalia R, Abdelaziz M, Puteri MU, Hwang J, Anwar F, Watanabe Y, Kato M. TMEPAI/PMEPA1 inhibits Wnt signaling by regulating β-catenin stability and nuclear accumulation in triple negative breast cancer cells. Cell Signal. pii: S0898-6568(19)30059-2, 2019. doi: 10.1016/j.cellsig.2019.03.016. PMID: 30890370
Abdelaziz M, Watanabe Y, Kato M. PMEPA1/TMEPAI knockout impairs tumor growth and lung metastasis in MDA-MB-231 cells without changing monolayer culture cell growth. J Biochem. 165(5): 411-414, 2019. doi: 10.1093/jb/mvz022. PMID: 30873542
Wardhani BWK, Puteri MU, Watanabe Y, Louisa M, Setiabudy R, Kato M. Decreased sensitivity of several anticancer drugs in TMEPAI knockout triple-negative breast cancer cells. Medical J. Indonesia 28: 110-115, 2019. doi: 10.13181/mji.v28i2.2687
Puteri MU, Watanabe Y, Wardhani BWK, Amalia R, Abdelaziz M, Kato M. PMEPA1/TMEPAI isoforms function via its PY and Smad interaction motifs for tumorigenic activities of breast cancer cells. Genes Cells, 25(6): 375-390, 2020. DOI: 10.1111/gtc.12766 PMID: 32181976
Research papers about TSC22 Family
Zheng L, Suzuki H, Nakajo Y, Nakano A, Kato M. Regulation of c-MYC transcriptional activity by transforming growth factor-beta 1-stimulated clone 22. Cancer Sci. 109:395-402, 2018. doi: 10.1111/cas.13466. Epub 2018 Jan 9. PMID: 29224245
Tran SPT, Hipolito CJ, Suzuki H, Xie R, Huynh Dam KT, ten Dijke P, Terasaka N, Goto Y, Suga H, Kato M. Generation of non-standard macrocyclic peptides specifically binding TSC-22 homologous gene-1. Biochem Biophys Res Commun. 516(2): 445-450, 2019. doi: 10.1016/j.bbrc.2019.06.035. PMID: 31227214
Zhang X, Koga N, Suzuki H, Kato M. Promotion of cellular senescence by THG-1/TSC22D4 knockout through activation of JUNB. Biochem Biophys Res Commun. 522(4):897-902, 2020. doi: 10.1016/j.bbrc.2019.11.145. PMID:31806366
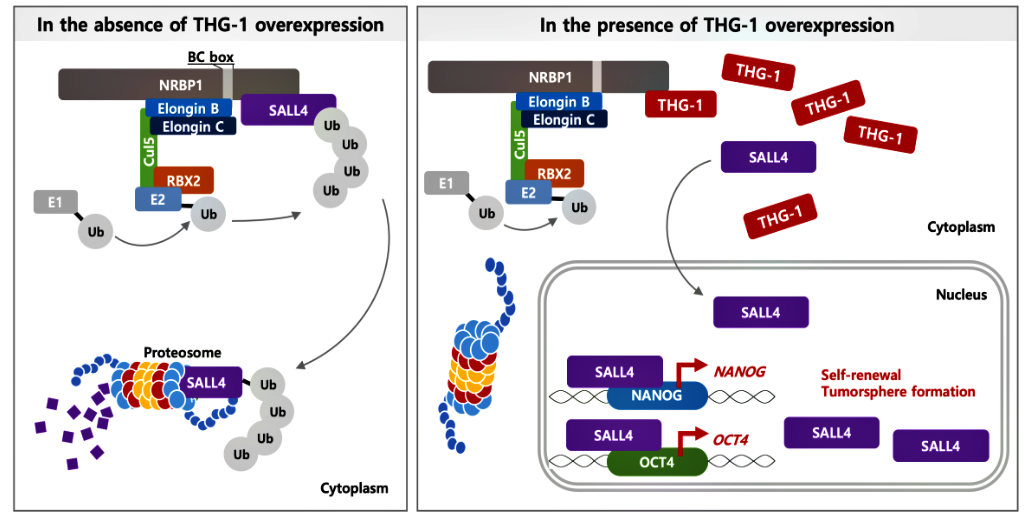
Hwang J, Haque MA, Suzuki H, ten Dijke P, Kato M. THG-1 suppresses SALL4 degradation to induce stemness genes and tumorsphere formation through antagonizing NRBP1 in squamous cell carcinoma cells. Biochem Biophys Res Commun. 523(2):307-314, 2020. doi:10.1016/j.bbrc.2019.11.149. PMID:31864704
Research Division Leader
Professor Mitsuyasu Kato

Research Division Leader
See Associated External Research Website